Mitiglinide
 | |
| Clinical data | |
|---|---|
| Trade names | Glufast |
| AHFS/Drugs.com | International Drug Names |
| Routes of administration | By mouth (tablets) |
| ATC code | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
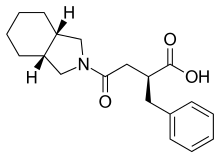
| Formula | C19H25NO3 |
| Molar mass | 315.413 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Mitiglinide (INN,[1] trade name Glufast) is a drug for the treatment of type 2 diabetes.[2]
Mitiglinide belongs to the meglitinide (glinide) class of blood glucose-lowering drugs and is currently co-marketed in Japan by Kissei and Takeda. The North America rights to mitiglinide are held by Elixir Pharmaceuticals. Mitiglinide has not yet gained FDA approval.
Pharmacology
[edit]Mitiglinide is thought to stimulate insulin secretion by closing the ATP-sensitive potassium KATP channels in pancreatic β cells.
Dosage
[edit]Mitiglinide is delivered in tablet form.
References
[edit]- ^ "International Nonproprietary Names for Pharmaceutical Substances (INN). Recommended International Nonproprietary names (Rec. INN): List 40" (PDF). World Health Organization. p. 187. Retrieved 10 November 2016.
- ^ Malaisse WJ (October 2008). "Mitiglinide: a rapid- and short-acting non-sulfonylurea insulinotropic agent for the treatment of type 2 diabetic patients". Expert Opinion on Pharmacotherapy. 9 (15): 2691–8. doi:10.1517/14656566.9.15.2691. PMID 18803455. S2CID 73318104.
External links
[edit]- Elixir Pharmaceuticals — website of the U.S. rights holder for mitiglinide.


 French
French Deutsch
Deutsch