 | Moderna COVID-19 vaccine (redirect from MRNA-1273) mRNA-1273/background/2021.1. Scholia has a profile for mRNA-1273 vaccine (Q87775025). Wikimedia Commons has media related to MRNA-1273. Wikinews... 165 KB (13,017 words) - 17:59, 27 April 2024 |
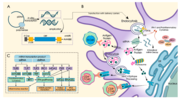
 | Solid lipid nanoparticle (redirect from MRNA-lipid nanoparticle) a complex of plasmid or linear DNA and lipids Targeted drug delivery mRNA-1273, from Moderna, uses LNPs BNT162b2, from BioNTech/Pfizer, uses LNPs Saupe... 23 KB (2,629 words) - 15:37, 19 March 2024 |
 | ; Hernán, Miguel A. (2022-07-01). "Comparative Safety of BNT162b2 and mRNA-1273 Vaccines in a Nationwide Cohort of US Veterans". JAMA Internal Medicine... 16 KB (1,510 words) - 19:37, 11 April 2024 |
PMID 30785039. Safety and Immunogenicity Study of 2019-nCoV Vaccine (mRNA-1273) for Prophylaxis of SARS-CoV-2 Infection (COVID-19), clinicaltrials.gov... 7 KB (628 words) - 18:17, 27 March 2024 |
 | the Moderna COVID‑19 vaccine candidate, and in December, the vaccine, mRNA-1273, was issued an emergency use authorization in the United States. In 2022... 23 KB (1,818 words) - 19:04, 19 April 2024 |
COVID-19 vaccine (redirect from MRNA-lipid nanoparticle COVID-19 vaccines) mRNA-1273/background/2021.1. Archived from the original on 13 June 2021. Retrieved 24 July 2021. "Background document on the mRNA-1273 vaccine... 192 KB (21,930 words) - 19:29, 15 April 2024 |
 | which biotechnology company Moderna used as the basis for the vaccine mRNA-1273, the first COVID-19 vaccine candidate to enter phase I clinical trials... 30 KB (2,822 words) - 17:59, 15 February 2024 |
Nucleoside-modified messenger RNA (redirect from Nucleoside-modified mRNA) developed by the cooperation of BioNTech/Pfizer (BNT162b2), and by Moderna (mRNA-1273). The zorecimeran vaccine developed by Curevac, however, uses unmodified... 24 KB (2,503 words) - 11:29, 22 February 2024 |
 | 2020, Moderna struck a manufacturing deal with Lonza to produce its (mRNA-1273) COVID-19 vaccine active ingredient while expanding sterile production... 18 KB (1,817 words) - 19:54, 20 March 2024 |