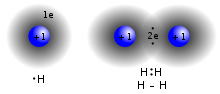
A three-center two-electron (3c–2e) bond is an electron-deficient chemical bond where three atoms share two electrons. The combination of three atomic... 5 KB (688 words) - 12:00, 26 March 2023 |
 | A 4-center 2-electron (4c–2e) bond is a type of chemical bond in which four atoms share two electrons in bonding, with a net bond order of 1⁄2. This type... 4 KB (430 words) - 10:40, 24 August 2023 |
The 3-center 4-electron (3c–4e) bond is a model used to explain bonding in certain hypervalent molecules such as tetratomic and hexatomic interhalogen... 11 KB (1,215 words) - 21:19, 20 March 2024 |
are two types of three-center bonds: Three-center two-electron bond, found in electron-deficient compounds such as boranes Three-center four-electron bond... 276 bytes (72 words) - 06:04, 20 March 2021 |
 | VSEPR theory (redirect from Valence shell electron pair repulsion) two lone pairs and two bond pairs. The four electron pairs are spread so as to point roughly towards the apices of a tetrahedron. However, the bond angle... 45 KB (4,038 words) - 13:09, 14 April 2024 |
 | Lone pair (redirect from Free electron pair) pair of valence electrons that are not shared with another atom in a covalent bond and is sometimes called an unshared pair or non-bonding pair. Lone pairs... 23 KB (2,947 words) - 11:17, 31 January 2024 |
"Exceptionally Long (2.9 Å) C–C Bonds between [TCNE]− Ions: Two-Electron, Four-Center π*–π* C–C Bonding in π-[TCNE]22−". Angewandte Chemie International Edition... 11 KB (1,125 words) - 00:59, 15 March 2024 |