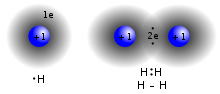
coordinate covalent bond, also known as a dative bond, dipolar bond, or coordinate bond is a kind of two-center, two-electron covalent bond in which the two... 10 KB (1,312 words) - 22:24, 19 April 2024 |
1,3-dipolar cycloaddition is a chemical reaction between a 1,3-dipole and a dipolarophile to form a five-membered ring. The earliest 1,3-dipolar cycloadditions... 65 KB (6,426 words) - 14:32, 7 March 2024 |
 | and only two from carbon, one bonding orbital is occupied by two electrons from oxygen, forming a dative or dipolar bond. This causes a C←O polarization... 72 KB (7,521 words) - 14:03, 12 April 2024 |
 | towards the oxygen atom. On the other hand, in the structure with a dipolar bond, the charge is localized as a lone pair on the oxygen. Typically metal... 17 KB (4,425 words) - 13:07, 17 April 2024 |
convection cooling curve coordinate chemistry coordinate covalent bond See dipolar bond. coordination complex A chemical compound consisting of a central... 170 KB (18,187 words) - 06:57, 23 February 2024 |
 | The residual dipolar coupling between two spins in a molecule occurs if the molecules in solution exhibit a partial alignment leading to an incomplete... 30 KB (3,545 words) - 00:55, 1 March 2024 |
Zwitterion (redirect from Dipolar zion) German Zwitter [ˈtsvɪtɐ] 'hermaphrodite'), also called an inner salt or dipolar ion, is a molecule that contains an equal number of positively and negatively... 10 KB (1,042 words) - 20:48, 2 April 2024 |
Azide-alkyne Huisgen cycloaddition (redirect from Huisgen 1,3-dipolar cycloaddition) The azide-alkyne Huisgen cycloaddition is a 1,3-dipolar cycloaddition between an azide and a terminal or internal alkyne to give a 1,2,3-triazole. Rolf... 21 KB (2,635 words) - 10:06, 9 April 2024 |