Clinical study design is the formulation of trials and experiments, as well as observational studies in medical, clinical and other types of research... 10 KB (1,030 words) - 17:15, 26 February 2024 |
 | Clinical trials are prospective biomedical or behavioral research studies on human participants designed to answer specific questions about biomedical... 112 KB (12,658 words) - 10:41, 24 April 2024 |
For drug development, the clinical phases start with testing for drug safety in a few human subjects, then expand to many study participants (potentially... 32 KB (3,806 words) - 19:25, 5 January 2024 |
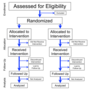 | Randomized controlled trial (redirect from Randomized clinical trial) policies requiring mandatory clinical trial registration as a prerequisite for publication. One way to classify RCTs is by study design. From most to least common... 88 KB (9,887 words) - 17:07, 24 April 2024 |
important in health care. In a randomized clinical trial, the subjects are randomly assigned to different arms of the study which receive different treatments... 7 KB (849 words) - 18:16, 28 November 2023 |
procedures are designed for the prevention, treatment, diagnosis or understanding of disease symptoms. Clinical research is different from clinical practice:... 9 KB (989 words) - 21:52, 9 March 2024 |
(mathematics) Climate ensemble Climograph Clinical significance Clinical study design Clinical trial Clinical utility of diagnostic tests Cliodynamics... 87 KB (8,293 words) - 21:10, 26 March 2024 |



