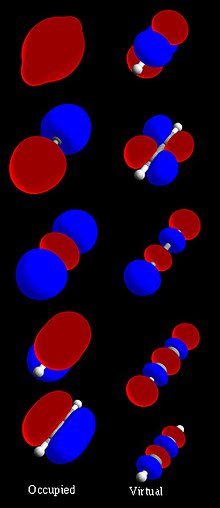
| chemical bonds. Antibonding molecular orbitals (MOs) are normally higher in energy than bonding molecular orbitals. Bonding and antibonding orbitals form... 7 KB (880 words) - 07:57, 10 May 2023 |
 | bonded atoms or passing through the bonded atoms. The corresponding antibonding, or σ* orbital, is defined by the presence of one nodal plane between... 8 KB (909 words) - 16:39, 8 November 2023 |
 | atoms, and no nodal planes between the bonded atoms. The corresponding antibonding, or π* ("pi-star") molecular orbital, is defined by the presence of an... 7 KB (816 words) - 06:21, 24 December 2023 |
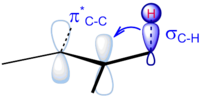
 | electrons back from a (different) filled d-orbital into the empty π* antibonding orbital. Both of these effects tend to reduce the carbon-carbon bond... 3 KB (318 words) - 21:43, 8 November 2022 |
dibromide adopts the trans configuration. For maximum overlap of the C–Br σ* antibonding molecular orbital (the LUMO, shown to the right in red) and the nucleophile... 7 KB (801 words) - 18:25, 20 April 2021 |
the difference between the numbers of electron pairs in bonding and antibonding molecular orbitals. Bond order gives a rough indication of the stability... 9 KB (1,239 words) - 05:21, 12 February 2023 |