Adalimumab, sold under the brand name Humira and others, is a disease-modifying antirheumatic drug and monoclonal antibody used to treat rheumatoid arthritis... 91 KB (6,940 words) - 04:46, 30 April 2024 |
effects can be achieved with a monoclonal antibody such as infliximab, adalimumab, certolizumab pegol, and golimumab, or with a circulating receptor fusion... 34 KB (3,827 words) - 06:58, 22 April 2024 |
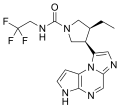 | superior to adalimumab for ACR50, DAS28CRP≤3.2, ΔPain and ΔHAQDI. At week 26, more participants on upadacitinib vs placebo or adalimumab achieved low... 46 KB (4,534 words) - 06:57, 1 April 2024 |
proceeding with this at present." Biosimilars available in Australia include adalimumab, bevacizumab, enoxaparin, epoetin lambda, etanercept, filgrastim, follitropin... 95 KB (6,479 words) - 21:14, 31 March 2024 |
in Forbes Global 2000 was 74. The company's primary product is Humira (adalimumab) ($14 billion in 2023 revenues, 27 percent of total), administered via... 39 KB (3,327 words) - 10:40, 11 May 2024 |
(disease-modifying antirheumatic drugs), monoclonal antibodies, such as infliximab and adalimumab, the TNF inhibitor etanercept, and methotrexate for moderate to severe... 15 KB (1,474 words) - 08:38, 1 May 2024 |
comprising four clinical trials which compared risankizumab to ustekinumab, adalimumab and placebo in the indication of plaque psoriasis. The results of these... 13 KB (969 words) - 19:29, 20 December 2023 |


