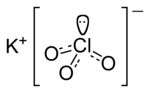
Chlorate is the common name of the ClO− 3 anion, whose chlorine atom is in the +5 oxidation state. The term can also refer to chemical compounds containing...
8 KB (740 words) - 13:04, 24 April 2024
Potassium chlorate is a compound containing potassium, chlorine and oxygen, with the molecular formula KClO3. In its pure form, it is a white crystalline...
15 KB (1,528 words) - 07:10, 13 May 2024
Sodium chlorate is an inorganic compound with the chemical formula NaClO3. It is a white crystalline powder that is readily soluble in water. It is hygroscopic...
19 KB (1,810 words) - 19:08, 22 May 2024
Ammonium chlorate is an inorganic compound with the formula NH4ClO3. It is obtained by neutralizing chloric acid with either ammonia or ammonium carbonate...
4 KB (312 words) - 14:49, 24 January 2024
Calcium chlorate is the calcium salt of chloric acid, with the chemical formula Ca(ClO3)2. Like other chlorates, it is a strong oxidizer. Calcium chlorate is...
5 KB (402 words) - 00:32, 21 May 2024
Sodium hypochlorite (redirect from Sodium chlorate(I))
sodium chlorate and sodium chloride: 3 NaOCl(aq) → 2 NaCl(aq) + NaClO3(aq) This reaction is exploited in the industrial production of sodium chlorate. An...
55 KB (5,761 words) - 14:08, 24 May 2024
Sodium chlorite (redirect from Sodium chlorate(III))
similar to the well known sodium chlorate: methemoglobinemia, hemolysis, kidney failure. A dose of 10-15 grams of sodium chlorate can be lethal. Methemoglobemia...
22 KB (1,815 words) - 00:11, 3 May 2024
Silver chlorate (AgClO3) forms white, tetragonal crystals. Like all chlorates, it is water-soluble and an oxidizing agent. As a simple metal salt, it is...
3 KB (187 words) - 18:26, 23 September 2023
Flash powder (section Aluminium and chlorate)
mixture to increase the sensitivity. Early formulations used potassium chlorate instead of potassium perchlorate. Flash powder compositions are also used...
15 KB (2,048 words) - 18:10, 26 March 2024
Chloric acid (redirect from Hydrogen chlorate)
Chloric acid, HClO3, is an oxoacid of chlorine, and the formal precursor of chlorate salts. It is a strong acid (pKa ≈ −2.7) and an oxidizing agent. Chloric...
4 KB (243 words) - 15:24, 3 November 2023